Spring, 2015
Have you ever wondered why foods contain trans fats, or why dough needs so much salt, or why cheese tastes so good?
Dr. David Pink at St. Francis Xavier University is a theoretical physicist working on edible oils, with the intent of making them healthier for people to eat. Food companies will have to replace certain trans fats in their products and the big question is “by what can they be replaced?” The problem arises from the fact that (solid) trans fats help create liquid oil-binding structures in products such as shortenings. Without their “Oil Binding Capacity”, shortenings and similar products just won’t work. But, industrially-manufactured trans fats created via hydrogenation have been shown to damage human health and the US Food & Drug Administration (USFDA) will require producers to cease using them. Accordingly, to replace them, we need alternatives that are inexpensive, edible and behave the same way as trans fats so that consumers won’t be able to tell the difference.
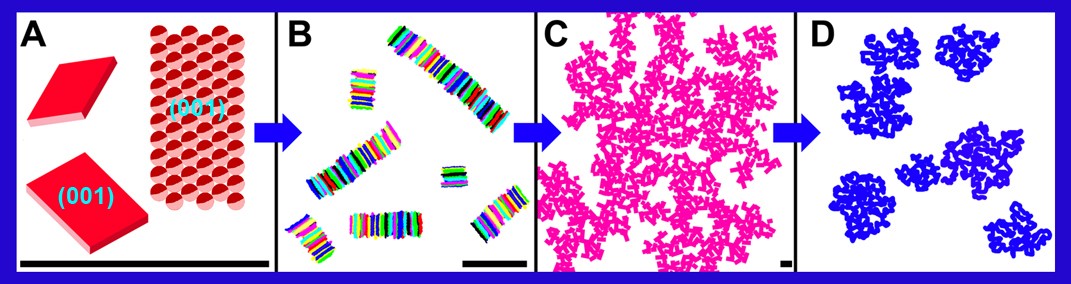
Working in collaboration with Dr. Alejandro Marangoni, Professor of Food Science at the University of Guelph, the process begins with trying to understand what solid fats structures arise in edible oils and why they trap the oil so as to exhibit the desired Oil Binding Capacity. Without understanding those fundamental things, researchers can only make, at best, educated guesses as to what ingredient can be used to successfully replace them – an inefficient way to try to solve a multimillion dollar problem.
Instead, Pink began by modelling crystalline nanoplatelets, the most stable small solid fats in the oils, then simulated their interactions to study them. Because of the models’ complexity – edible oils involve many components – he had to make use of computer simulations, working with his Research Associate, Bonnie Quinn, and using ACENET. The goal was to predict what structures develop in edible oils. Pink says, “If it wasn’t for ACENET, I wouldn’t even be able to begin.”.
Pink predicted the experimental structures which arise in systems using ultra-small angle X-ray scattering (USAXS). He also predicted the existence of stacks of crystalline nanoplatelets, called TAGwoods. His predictions were all confirmed by his PhD student. He then made predictions about solid fats which become coated with semi-liquid oil components. He, Dr. Shajahan Razul of ACENET and Pink’s undergraduate student carried out atomic scale molecular dynamics simulations and showed that Pink’s assumptions about coated solid fats were justified and that they will indeed bind oil. Subsequent projects will address how larger structures get formed, so that he can begin searching for replacements for trans fats. One such project will probe the dynamics of edible oils using dissipative particle dynamics, a simulation technique that can study larger scale systems over longer timescales.
In recent months, Pink has branched out. With Dr. Marangoni’s leadership and an industry collaboration, he is modelling cheeses to discover the essential aspects of their structures.
Pink is also modelling dough. Bread makers will be required to reduce the amount of salt and this can make the dough sticky. Working with Dr. Erzsebet Papp-Szabo, who provides input about the chemistry of dough, he has just finished developing models and has some preliminary results.
Now in his 70s, by most people’s standards, David Pink should be retired. “But,” he said, “I’m simply having too much fun working with Alejandro, with my other colleagues and with my students.” And he is pleased that his work has been recognized by the 2015 Distinguished Service/Outstanding Achievement Award from the Edible Applications Technology division of the American Oil Chemists’ Society.